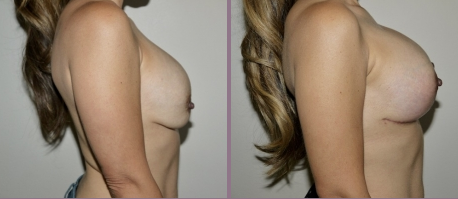
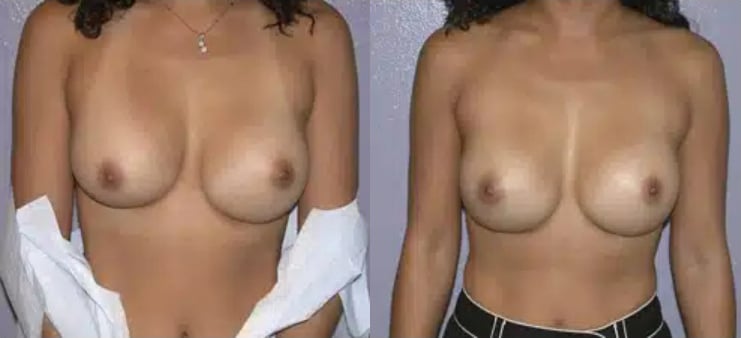
Board Certified Female Plastic Surgeon Specializing in Breast Augmentation Revision & Implant Removal
Breast implants are very durable and should last a long time, but they’re not expected to be lifetime devices. At some point, your implants may wear out and require replacement. This is natural and should be expected whenever you contemplate undergoing breast augmentation.
That doesn’t mean you are committed to breast surgery on a regular basis. If everything looks and feels good and there is no evidence of rupture, you can keep your implants for many years. That being said, at some point in time, you will most likely need to remove your old breast implants and replace them with a new set.
It’s important to realize that the removal and replacement of breast implants is often desired for cosmetic reasons rather than ruptures or other complications. Whether you're unhappy with the outcome of a previous breast augmentation or you’ve experienced an implant rupture, you have several options available to you. Dr. Hayley Brown has extensive experience performing breast augmentation revision procedures, and she can devise a treatment plan that will deliver the exceptional results you deserve.
You can receive individualized treatment recommendations by speaking with Dr. Brown in person. Please call 702-260-7707 today to schedule a consultation. We serve patients in Las Vegas and Henderson, Nevada.
- Is Breast Implant Removal and Replacement Right for Me?
- Common Cosmetic Reasons for Breast Implant Removal and Replacement
- Breast Augmentation Revision to Correct Poor Outcomes from a Previous Procedure
- What Does Capsular Contracture Look Like?
- Breast Implant Rupture
- Surgical Procedures to Remove and Replace Breast Implants
- Breast Implant Removal with Lift
- Is Breast Implant Removal Painful?
- How Much Does Breast Implant Removal Cost?
- Does Insurance Pay for Breast Implant Removal?
- Indications for capsulectomy and en bloc technique
- Contact our Las Vegas Plastic Surgeon
- The Aesthetic Society Advisory on Breast Implant Illness
The reasons for breast implant removal are as varied as the reasons for undergoing breast augmentation. It is a personal decision that each woman should make to satisfy her own life and body goals. As you experience more time with your breast implants, you may wish to have them removed at some point. Removal of breast implants is more common than many women believe.
You may identify with these common reasons for considering breast implant removal:
- Dissatisfaction with your appearance – After several years with implants, some women just want to return to their smaller curves. Other women were never truly satisfied with the results of their first breast augmentation. All women can be affected by age, pregnancy, menopause, and weight gain or loss. These factors will alter the appearance of your breast implants.
- Desire to increase physical activity – Larger breasts can be a hindrance to an active lifestyle. They can affect the range of motion of your arms when participating in sports such as golf or tennis. The additional weight in your breast region can also make running more challenging and in some cases, it can result in back pain.
- Deflation or capsular contracture – When implants rupture or the body has formed a hard cavity around the implant, it can lead to an abnormal appearance that can be corrected with implant removal or replacement.
- Styles have changed – The larger breast implants that were popular in the 80s and 90s do not suit the body trends of today. This can lead women with larger implants to feel self-conscious and yearn to return to smaller, shapelier breasts.
Common Cosmetic Reasons For Breast Implant Removal And Replacement
The following are some of the most common cosmetic reasons for breast implant removal and replacement.
Desire for a Larger Implant Size
Desire for a Smaller Breast Implant
This is less common, but it does occur on occasion. Some implant sizes look large on small frames and in some instances, women complain of difficulty buttoning tops or jackets after breast augmentation. Some women simply admit to making a poor choice of size, not realizing how big they would look, or they feel like they look heavy with larger breasts. Occasionally women complain of back, neck and shoulder pain from large implants.
If a woman has good skin tone and no sagging issues, a conservative downsize may be appropriate. Keep in mind that going much smaller can lead to some skin redundancy and sagging, that could require breast lift surgery. In many cases, it is not possible to downsize an implant as there is no way to shrink a pocket in all dimensions. If a woman desires a noticeable size decrease, usually implant removal with breast lift is the best option.
Switching from Saline to Silicone Breast Implants
This involves removing saline implants for the more natural feel of silicone implants. There was a 14-year ban on silicone breast implants from 1992-2006, and many women who received saline implants during this time have opted for surgery to switch their device to the newer generation soft and natural feeling silicone breast implants. Patients who have the old leaky silicone devices may also opt to have their implants replaced with the newer generation cohesive gel devices.
Augmentation Mastopexy Surgery to Address Sagging Issues
Breast implants add volume to breasts, but they do not treat sagging, defy gravity, or lift a breast. The added volume and weight from breast implants can aggravate sagging over time, especially in patients with poor skin tone and loose skin characterized by stretch marks.
Surgery to correct this issue involves removal and replacement of implants and a simultaneous breast lift. Implant size can go up or down, depending on patient desires.
Explantation (Implant Removal)
Some women are just “over” having large breasts. Some women have regrets about their initial decision to undergo breast augmentation in the first place. A certain subset of women notice increases in bra size as they age. Another subgroup of patients has aged and they are simply unconcerned about their breast size or appearance.
Explantation can be done with or without breast lift surgery. Most woman have a simultaneous breast lift to reshape the breast to a rounder, perkier and more youthful contour. Without this additional procedure, the breasts can look saggy and deflated with risks of depressions and contour irregularities.
Breast Augmentation Revision To Correct Poor Outcomes From A Previous Procedure
When breast augmentation is performed by an experienced board certified plastic surgeon, complications are rare. However, as with all surgical procedures, there’s still some risk that complications might occur. In these situations, you may be able to correct the poor outcome with a breast augmentation revision procedure.
Capsular Contracture
Capsular contracture is a condition that results in increased firmness of the breast capsule. A thin capsule or scar tissue sac is a normal response to any foreign object placed within the body. Usually the scar is not seen or felt.
However, everyone heals differently. Some patients form thicker capsules than others. Moderate to severely thick capsules squeeze the implant, making it feel uncomfortably firm. In these cases it can change the breast contour or cause pain.
Some potential risk factors for the development of capsular contracture include:
- The way the surgery is performed
- Using a periareolar or transaxillary incision
- Spillage of blood in the pocket during the surgery
- Hematoma (bleeding)
- Subclinical infection
- A patient’s inherent response to a foreign object placed in their body
Implant Malposition
Examples of implant malposition include:
- Lateral displacement (implants falling out to the side, usually from gravitational forces)
- Bottoming out (implants sit too low)
- One or both implants sit too high
- Symmastia (implants are too close, creating a “uniboob” appearance)

Rippling
Rippling can be associated with thin patients or larger implants. Pressure-related thinning of surrounding breast tissue and flattening of the chest wall can occur over time, especially with larger implant sizes. Larger implants often result in more tissue stretch, thinning, rippling, and sagging issues over time.
All of these problems are difficult to correct in the future, so choose your initial size wisely. Bigger isn’t always better.
It’s important to understand that rippling does not mean rupture, although increased folds can be a symptom of rupture. Rippling occurs from thinning. Any implant will show if your tissues are thin enough.
Contour Irregularities
Contour irregularities can affect the lower breast fold. These deformities and “double bubble signs” can occur for several reasons. If you start to experience sagging and the implant was placed under the muscle, you may be able see the lower round implant edge behind the breast, and the natural breast fold above it. It looks like a double crease.
Sagging Issues
Early aggravated sagging can occur in breast augmentation patients whose tissues are loose, lax, and stretchable. This can occur in predisposed women with poor skin tone, where their tissues simply do not support the weight of the implant. Whether the implant is placed under or over the muscle is not always relevant. Surgery usually requires implant downsize and a breast lift.
What Does Capsular Contracture Look Like?
Mild capsular contracture may not be externally visible. Your breast will look normal, but feel slightly firm to the touch and there may be a slight lifting effect to the breast. Severe capsular contracture, on the other hand, may result in a harder feeling breast along with increased upper pole fullness and changes in breast shape. In some cases, the implant itself may shift noticeably higher, causing asymmetry between your breasts.
Breast Implant Rupture
Most shell failures in a breast implant are due to wear and tear, not from trauma to the breast region. A tear or hole in the implant shell is called a rupture. If you have saline breast implants, the opening can allow saline to escape from the shell, and you may experience a size decrease as the sterile saline is immediately or sometimes gradually absorbed.
Ruptures with silicone implants are different. Newer generation silicone implants are cohesive or form stable, so the silicone tends to stay within the shell and you may have no symptoms of shell rupture. This is called a "silent rupture." The FDA recommends that women who receive newer generation cohesive silicone implants have MRIs of their breasts at certain intervals to make sure the shell is intact.
The implant shell will not degrade in your body. The risk of implant rupture increases with implant age. Rupture rates increase 1% per year, so the risk is roughly 10% at 10 years.
Ruptures of both saline and silicone implants have no impact on your health. The FDA has determined that there is no danger from internal exposure to silicone. There is no relationship of silicone exposure to autoimmune disease, cancer, rheumatologic disease, connective tissue disease or illness. Therefore, removal and replacement surgery is performed at your convenience.
Surgical Procedures To Remove And Replace Breast Implants
Surgery to remove and replace implants ranges from straightforward and simple to complicated and expensive. It depends on the situation at hand, your goals and desires, your tissue characteristics, and basically what there is to work with.
Secondary surgery in general is usually more complicated than primary breast augmentation surgery. Reasons for this include:
- The presence of scar tissue, old pockets and capsules
- Thinning of overlying tissue, stretched skin, sagging
- Changes in blood supply to the nipple and overlying skin envelope
These surgeries often take longer than the initial surgery and are more expensive due to their complexity. It’s important to understand the financial obligations associated with revision procedures.
In general, Dr. Brown advises all patients with excellent results to avoid revision surgery unless it’s truly necessary. She also discourages surgery for small size changes or in women who want to go with implants that are much too large for their frame.
All women undergoing breast augmentation need to expect implant maintenance surgery. The majority of patients have excellent short-term and long-term results with no complications. However, expect the need for a revision procedure at some point in time, with the typical reason being cosmetic in nature.
Breast Implant Removal With Lift
Once implants have been removed, it’s important to consider the appearance of your chest after the procedure. For many women, a breast lift combined with implant removal is an ideal combination of procedures to help achieve more desirable results.
Many women notice that after breast implants are removed and the breast loses that extra volume, excess skin remains. This stretching of the skin can cause your chest to appear droopy, saggy or deflated.
A breast lift is an ideal procedure to help counteract the unwanted aesthetic effects of breast implant removal. Sometimes Dr. Brown may recommend an alternative procedure such as an implant removal with replacement depending on your specific goals.
In general, a breast lift will be the best option if you are removing your implants because you:
- Have health concerns
- Want to look youthful, thinner, and pursue an active lifestyle
- Want to continue to look great in clothes and swimsuits with or without wearing a bra
- Have experienced a gain in breast tissue or sagging over the years
- Simply no longer wish to have implants
- Have back pain, neck pain, or bra strap grooving or discomfort from large implants
Alternatively, you might benefit from breast implant removal and replacement if you are removing your implants because:
- You are uncomfortable with how your current implants feel
- You are unhappy with the overall look of your current implants
- You are not satisfied with the size or position of your implants
Is It Painful To Have Breast Implants Removed?
It is not any more painful to have breast implants removed than it is to have them placed. Depending on how extensive and complex your revision surgery is, you may need to take anywhere from a few days to a week off from work in order to rest and recover. We'll prescribe you medications to help manage any pain or discomfort you may feel during your recovery.
How Much Does Implant Removal Cost In Las Vegas?
Breast implant removal at Desert Hills Plastic Surgery Center starts at $9,000. When combined with a breast lift, the price starts at $20,000. Revisionary surgery to remove and replace implants begins at $15,000 depending on the complexity of the surgery. Removal and replacement with a lift starts at $20,000. After your consultation with Dr. Brown, we will make sure you know what the cost is and your options for payment.
Indications For Capsulectomy And En Bloc Technique
Capsulectomy means removal of the scar around your implant at the time of implant removal. This surgical procedure may or may not be required at the time of your implant removal breast lift surgery. There are many variables that need to be considered when determining the need for capsulectomy and the final decision is often made intraoperatively, based on the appearance of the capsule and tissues within the breast. In many cases, the capsular tissue is not visible as a distinct entity. It can simply look like the surrounding tissues and it can be technically difficult and unnecessary to peel it away from the surrounding structures. In these cases, removal can be harmful, risking damage to deeper structures, getting into the chest cavity (pneumothorax), causing bleeding and even causing skin and nipple damage or loss. When a woman has a capsular contracture, the surrounding scar around the implant is thick, white, hard, firm and sometimes calcified like an eggshell. In these cases the capsular tissue is removed with the implant. An “en bloc” technique means removing the implant and its surrounding capsule as a unit, ideally without entering the capsule during the dissection around it. This technique is used for cancer treatments, and may be an efficient way to clean out ruptured silicone implants with a severe surrounding capsular contracture. When the capsule is hard like a rock, it is fairly easy to get around it and remove it as a whole allowing for an easy clean-up of a ruptured implant while minimizing surrounding tissue exposure to silicone during removal.
In recent times, the en bloc technique of implant removal has become very familiar to women considering breast implant removal. There is a subset of women who want their implants removed for health concerns. Some of these women have a constellation of symptoms with no apparent etiology, also called "breast implant illness", and they hope that implant removal will improve their health. The internet and social media is full of unsubstantiated claims about the presumed advantages of en bloc technique of breast implant removal in optimizing a woman’s health. Keep in mind, that at this time, none of this has been proven in the scientific literature. It is unclear why some women feel better after their implants are removed. Studies have shown that improvement in symptoms is unrelated to whether or not the capsule is removed. In addition, there is no relationship of breast implants or silicone exposure to any autoimmune condition, cancer or any disease and that ruptures of saline and silicone implants are not dangerous or a health risk. Regardless, it is understandable that a woman suffering from health issues wants to do whatever she can to possibly feel better.
What women need to know, is that it may not be possible or safe to have en bloc removal of a breast implant and its associated capsule, especially in those considering simultaneous breast lift surgery and those without a capsular contracture.
When a woman has soft feeling implants (which is normal), the capsular tissue can be thinned out and indistinct as an independent layer. It looks like a see-through membrane that is filmy, insubstantial, and delicate like a cobweb and appears fused and continuous with the surrounding tissues. So what is this capsular tissue? It is your tissue, a barely perceptible layer of scar that is benign and a source of blood supply to overlying tissues. It is not dangerous, malignant, or foreign. There are certain scenarios where it can be technically impossible and unnecessary to remove all of the capsule en bloc and the risks of removal far outweigh the benefit:
- When the implants are under the muscle. The back layer of the capsule can be fused with the ribs with no safe plane under it. In these cases, attempted removal risks getting into the chest and lung causing air in the chest (pneumothorax), bleeding and damage to deeper structures.
- When implants have been place through an armpit approach, the capsule in the armpit can be technically challenging to reach and risky to remove because of critical structures in the armpit and risks for uncontrollable bleeding and damage to deeper structures.
- In patients with very thin tissues and history of rippling (visibility of an implant through thin tissues), removal of the capsule can cause overlying skin damage, nipple loss, skin loss, uncorrectable indentations, contour irregularities and poor aesthetics.
- Women with very large implants with loose, distended pockets or lateral displacement of the implant. In these cases, the pocket is so large that it can be impossible to get around this huge space.
- During simultaneous breast lift. Because the capsular tissue is a source of circulation to the overlying breast skin and nipple, its removal along with breast lift surgery may increase risks for skin and nipple loss from poor circulation.
- In general, when there is no capsular contracture and no implant rupture, the extensive dissection to remove capsule adds increased operating room time and increases all risks associated with surgery to include, bleeding, infection, anesthetic risks, permanent sensation changes, nipple and skin loss, contour irregularities and poor aesthetic outcome.
The most feasible situations for en bloc removal of a breast implant include:
- Severe capsular contractures with or without implant rupture
- Relatively small implants originally inserted using an areolar or inframammary fold incision
- Over the muscle placement
- Relatively thick breast tissue over the implant and under the skin
- any sort of malignancy would be an indication for en bloc technique
Currently there is insufficient evidence to state that the removal of breast implants and capsules with or without en bloc technique will alter the course or prevent autoimmune or other disorders alleged to be caused by breast implants. The removal of all or part of your capsule is dependent on many anatomic and intraoperative findings with a risk benefit analysis individualized for each patient so as not to increase risks for major complications.
Contact Our Las Vegas Plastic Surgeon
Dr. Hayley Brown understands the changes a woman's body can go through and how a previously satisfactory breast augmentation might lose its effectiveness over time. She has extensive experience performing breast augmentation revision procedures and will develop a customized treatment plan to address your unique needs and goals.
Please contact Desert Hills Plastic Surgery Center using the form on this page or call 702-260-7707 today to schedule your consultation. We serve patients in Las Vegas, Henderson and the surrounding areas of Nevada.
The Aesthetic Society Advisory On Breast Implant Illness
The following information first appeared on the ASAPS website on July 16, 2019
Breast implant illness (BII) is a subject that is increasingly on the minds of many of our patients. Because of low incidence and variable presentation, it will be some time before we understand the etiopathogenesis and best treatment for this condition.
The Aesthetic Society makes no attempt to speak for every Society member or for
plastic surgeons in general, but patients with this condition require and deserve the best possible care and must not be preyed upon by overly aggressive work-up and
treatment.The following is the Society’s current position on BII:
- There is currently no science to support genetic testing as a means of determining which patients may be susceptible to BII. The Aesthetic Society is in favor of, and the Society’s research arm, Aesthetic Surgery Education and Research Foundation, may provide financial support for, further research in this area.
- There is no evidence that capsulectomy is the treatment for BII. Further, there is no evidence that an en bloc capsulectomy is superior to precise capsulectomy or more limited forms of capsulectomy.
The Aesthetic Society will update the position on Breast Implant Illness as scientific data is accumulated.