Board Certified Female Plastic Surgeon Specializing in Breast Augmentation Surgery
Breast augmentation is consistently one of the most popular plastic surgery procedures performed every year. It’s an excellent option to help you achieve your desired appearance by improving the size and shape of your breasts.
Dr. Hayley Brown has 25 years of experience performing breast augmentation procedures. As a board-certified plastic surgeon, she understands the unique concerns women have in a way that most male surgeons can never truly grasp. This allows her to forge meaningful relationships with her patients.

Learn about your options and receive individualized breast augmentation recommendations by speaking with Dr. Hayley Brown in person. Please call 702-260-7707 today to schedule a consultation.
We proudly serve patients in Las Vegas and Henderson, Nevada.
- What is Breast Augmentation?
- Dr. Brown's Patient Discusses Her Breast Augmentation
- Benefits of Breast Augmentation
- Am I a Candidate for Breast Augmentation
- Your Customized Breast Augmentation Procedure
- How Much Does Breast Augmentation Cost in Las Vegas?
- Breast Implant Options
- Breast Implant Styles
- Breast Augmentation Incision Options
- Breast Implant Placement Options
- Breast Implant Sizing
- Recovery after Breast Augmentation
- Breast Augmentation with Lift
- Contact our Las Vegas Breast Augmentation Surgeon
What Is Breast Augmentation?
Breast augmentation, also called breast enhancement, is a procedure that uses saline or silicone breast implants to increase the size, volume, and contours of the breast.
Dr. Brown will evaluate the following factors when planning your breast augmentation:
- Your desired breast size
- Your natural breast size and the amount of natural breast tissue you have
- Your body frame and dimensions
- Whether you have breast asymmetry
- Whether you lead an active, athletic lifestyle
Dr. Brown's Patient Discusses Her Breast Augmentation
Benefits of Breast Augmentation
The greatest benefit of undergoing breast augmentation is an improved breast appearance and a more balanced body contour.
The procedure also offers a wide range of other aesthetic benefits besides size, including:
- Improved breast shape
- Correction of breast asymmetry
- Restoration of lost breast volume caused by weight loss, pregnancy and breastfeeding, or aging
- Improved breast projection
- More attractive cleavage
- Improved appearance in clothing, bathing suits, and lingerie
Am I a Candidate for Breast Augmentation?
If you would like to improve any aspect of your breast appearance, you are most likely a candidate for breast augmentation. In general, the best candidates for breast augmentation surgery want to achieve one or more of the following goals:
- Increase the size or improve the shape of their breasts
- Have better cleavage
- Correct breast asymmetry or a congenital deformity of the chest, such as tubular breast deformities or pointy-shaped breasts
- Restore lost breast volume
- Correct mild sagging issues (severe sagging issues are best corrected by a breast lift)
It's important to be in good health and have realistic expectations for the outcome of your procedure. You should not currently be pregnant or breastfeeding. You will also need to have enough natural breast tissue to cover the implant.
Smokers must be willing to quit prior to surgery and throughout the recovery period, since smoking can adversely impact healing and your results.
Due to age restrictions mandated by the FDA, you must be at least 18 for saline implants and at least 22 for silicone implants.
Your Customized Breast Augmentation Procedure
Breast augmentation is performed on an outpatient basis under general anesthesia. Dr. Brown uses the most advanced techniques to ensure that you get the look you're hoping for.
During your consultation, she will listen carefully to your concerns and goals and evaluate your body type. She'll then discuss your options in detail and work closely with you to develop a plan that will deliver your desired outcome.
The factors that will impact your breast augmentation plan include:
- Breast implant type
- Breast implant style
- Incision location
- Implant placement
- Implant size

How Do I Prepare for Breast Augmentation?
During your initial consultation for breast augmentation surgery, Dr. Brown will provide you with a list of instructions to follow to prepare for the procedure.
Before your surgery:
- Fill prescriptions for your pain medication.
- Purchase any medical supplies you may need, such as sterile gauze and the underwire bra specified at your pre-op visit.
- Find a trusted family member or friend to drive you to and from your surgery.
- Ask a family member or friend to take care of you in your initial days of recovery.
- Clean your house.
- Have on hand lots of prepared healthy food and electrolyte drinks, such as Gatorade.
- Set up your recovery area with pillows, blankets, a TV, and books to read.
Most patients begin to feel much better within a week of their surgery. Dr. Brown will be able to give you a timeline for your recovery period.
Will the Results of My Breast Augmentation Look Natural?
Breast augmentation is intended to look very natural. However, people who are intimately close to you will notice a difference in the shape of your breasts since they are most familiar with your body.
At first, the implants are high and firm, but as months pass, the implants will drop and settle. Individuals you meet in the future will not likely realize that you have undergone a breast augmentation procedure.
Alternatively, some patients desire a "fake" look, which can be achieved with larger and higher-profile implants.
How Long Will the Breast Augmentation Procedure Take?
The time required to complete a breast augmentation depends on your unique situation. Various techniques may be used to complete a breast augmentation procedure, and the type of method used can affect the length of your surgery.
Most surgeries may be completed in one to three hours. Dr. Brown can give you a customized estimate of the time that your breast augmentation will take.
How Soon After My Procedure Will I Be Able to See the Results?
You’ll immediately notice a difference in your breasts following your surgery. However, optimal results are apparent in two to four months. The implants need time to drop, settle, and soften into your natural breast pocket.
In most cases, your results can be long-lasting. However, implants are not expected to last forever. Most manufacturers offer a 10-year warranty for the implant shell.
Whether or not you've had surgery, it's normal to experience breast appearance changes from aging, gravity, weight fluctuation, pregnancy, and menopause. Maintenance surgery is therefore recommended for rupture or cosmetic reasons.
When Will I Be Able to Sleep on My Side After Breast Augmentation?
Following a breast augmentation, you should sleep on your back for three weeks in a slightly elevated position to prevent any pressure on the incisions and muscles surrounding the breast.
After three weeks, if sleeping on your back is uncomfortable, you may choose to sleep on your side with certain precautions. You’ll need to wear your underwire bra and place a soft pillow beneath the breasts to prevent any added pressure or shift of the implant as it heals.
How Much Does Breast Augmentation Cost?
At Desert Hills Plastic Surgery Center, the cost of breast augmentation starts at $8,000. One of the primary factors is whether saline or silicone implants are used. Saline breast implants are less expensive than silicone implants.
There may be other factors influencing the cost, such as whether any other procedures are combined with breast augmentation. For example, when a breast augmentation is combined with a breast lift, the cost starts at $15,000.
When you visit Dr. Brown and our staff for your consultation, we will provide you with a cost estimate after you meet with Dr. Brown. We do accept cash and credit cards. If you wish to apply for financing through Prosper or CareCredit, we can walk you through how to do so.
Breast Implant Options
At Desert Hills Plastic Surgery Center, Dr. Brown uses both saline and silicone implants from trusted FDA-approved companies like Mentor and Allergan. Each type has its own pros and cons, and Dr. Brown will help you decide which option best fits your body and goals.
Silicone Implants (MemoryGel® “Gummy Bear” Implants)
Silicone implants are filled with a soft gel that feels a lot like natural breast tissue. You may hear them called “gummy bear” implants because, like a gummy bear candy, they hold their shape if cut in half instead of leaking.
Benefits of silicone implants:
- Look and feel more natural
- Available in many sizes and shapes
- Less likely to ripple under the skin
- Hold their shape even if the shell is damaged
Considerations:
- Harder to detect if they rupture, so MRIs may be needed to check them
- Surgery is required if replacement is ever needed
Saline Implants
Saline implants are filled with a sterile saltwater solution after they’re placed in the breast.
Benefits of saline implants:
- Smaller incision for placement
- Size can be adjusted during surgery
- If they rupture, the body safely absorbs the fluid
Considerations:
- Feel less natural compared to silicone
- More likely to ripple
- Deflate quickly if they rupture, requiring revision surgery

Breast Implant Styles
You have several options when it comes to breast implant profiles and shapes:
- Moderate profile implants often provide the most natural look.
- Higher profile implants are designed narrower with more outward projection, providing a slightly rounder appearance. These implants work well for women with smaller or narrower frames who desire larger cup sizes than would typically accompany their frame.
- Moderate plus profile implants are a nice compromise between high and moderate.
- Extra high-profile implants appeal to women wanting a very large, round look.

Breast Augmentation Incision Options
Dr. Brown will recommend the incision type that best fits your body and goals. Incisions are usually small, about 3 cm for saline implants and a little longer for silicone, and the scars tend to fade and blend in over time.
Inframammary Incision (Under the Breast)
Made in the fold beneath the breast.
Benefits include:
- A hidden scar under the natural crease
- Gives the surgeon the most control for precise placement
- Lower risk of losing nipple sensation
- Less chance of interfering with breastfeeding
- Lower risk of scar tissue tightening around the implant (capsular contracture)
Periareolar Incision (Around the Nipple)
Made along the edge of the areola (the darker skin around the nipple).
Benefits include:
- Scar blends with the natural border of the areola
- Allows very precise implant placement
- Helpful for correcting inverted nipples
- Works well for women with darker skin tones (scar blends more easily)
- Can improve breast shape in women with tubular or pointy breasts
- Good option for women with mild to moderate sagging who want to avoid a breast lift
Breast Implant Placement Options
Dr. Brown offers three main ways to place breast implants. The best choice depends on your body type, natural breast tissue, and goals.
Submuscular Placement (Under the Muscle)
In this method, the implant is placed beneath the chest muscle. It’s often chosen by women with little natural breast tissue because the muscle provides extra padding. This placement tends to create a more natural look and feel while also reducing the chances of visible rippling.
Dual Plane Placement (Partially Under the Muscle)
With the dual plane approach, the upper part of the implant is placed under the muscle, while the lower part is left uncovered. This option works well for women with mild sagging or a lower breast position. It combines the advantages of both under-the-muscle and over-the-muscle placement, helping the lower breast look fuller while still maintaining some of the natural coverage provided by the muscle.
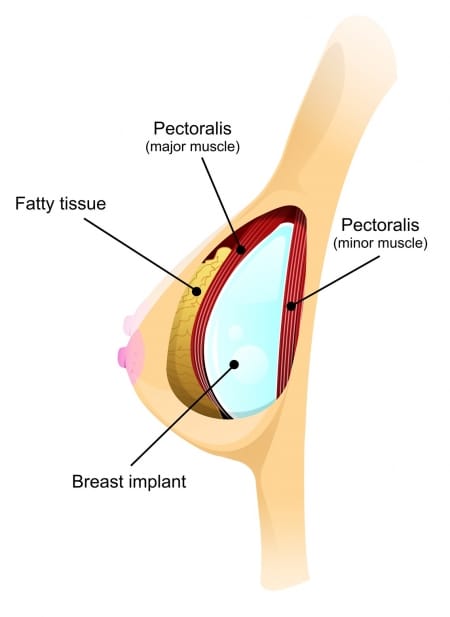
Subglandular Placement (Over the Muscle)
In subglandular placement, the implant sits behind the breast tissue but above the muscle. It is typically recommended for women who have thicker natural breast tissue and don’t need the added coverage of the muscle. This method can be especially helpful for women with mild to moderate sagging who want to avoid a breast lift, and it often creates a rounder look with fuller cleavage.
Breast Implant Sizing
Choosing the right implant size depends on several factors, including:
- Your height, weight, and chest shape
- How much natural breast tissue you already have
- The elasticity of your skin
- Your desired cup size
- Your activity level and lifestyle
Breast implants are measured in cubic centimeters (ccs), not cup sizes. This is because bra sizes vary from brand to brand, but ccs give a consistent way to measure volume.
General guide:
- Implants range from about 120 ccs to 850 ccs.
- Around 200 ccs usually equals about one cup size increase for smaller bra bands (32–34). Larger band sizes (36 and up) may need a little more volume to see the same change.
Dr. Brown will help you choose a size that fits your body and looks natural. For most women, a moderate size gives the best balance of shape, cleavage, and proportion. Women who are very athletic sometimes choose slightly smaller implants so their new breast size does not interfere with sports or fitness activities.
Recovery After Breast Augmentation
There will be some swelling and bruising, which will gradually subside over the first few weeks of your recovery. You may experience some discomfort, but this is easily managed by the oral pain medication and muscle relaxers prescribed by Dr. Brown.
Dr. Brown will provide you with a surgical bra after your procedure. As soon as 24 hours after surgery, you may change into a shapely underwire bra, size and style specified by Dr. Brown. This should be worn day and night for several months or until your implants drop and settle into their final position.
The underwire stabilizes the lower and outer breast pocket dimensions, leading to predictable implant position and central cleavage as the implants drop and settle with gravity. Bra support is encouraged long-term to decrease the risk of sagging. The larger your implants, the greater the risk of sagging due to gravity.
You should plan on taking one to two weeks off work while you recover. Refrain from upper body activity, heavy lifting, or fitness activities for three to four weeks. You can gradually resume exercise as tolerated after the first month of recovery.
Over time, your breast implants will soften, settle, and drop. This typically starts in two months, but the time frame can vary depending on your tissue tightness and skin tone. Mild changes in breast appearance can continue for up to six months after surgery.
Breast Augmentation With Lift
Sagging affects the majority of women at some point in their lives. Breast implants alone do not treat sagging breasts, especially in cases of moderate to severe sagging. If you would like to improve the size or shape of your breasts and also restore some perkiness, Dr. Brown may recommend breast augmentation with a breast lift for the most effective results.
Dr. Brown can use saline or silicone breast implants for this procedure. Silicone implants tend to feel more natural, making them the more popular option. Dr. Brown prefers submuscular implant placement for the majority of women who undergo breast augmentation with a lift.

Breast Augmentation for Athletic Women
Active women often have specific considerations when planning breast augmentation. Lower body fat and strong chest muscles can influence both implant size and placement. Larger implants may feel heavy during high-impact activity or upper body workouts, so many athletic patients prefer moderate sizes that enhance shape without interfering with performance.
Placement is also important. Submuscular or dual plane placement can provide additional coverage in women with minimal natural breast tissue, helping create a smooth contour. During the consultation, your training routine and long-term fitness goals should be part of the discussion so that your results align with your lifestyle.
How to Choose the Right Breast Implant Size for Your Body Type
Selecting the right implant size involves more than choosing a cup size. Implants are measured in cubic centimeters, and the best option depends on your chest width, skin elasticity, natural breast tissue, and overall proportions. An implant that suits one body type may look disproportionate on another.
Some women want subtle enhancement, while others prefer a fuller look. Trying implant sizers during your consultation can help you visualize different volumes on your frame. The goal is to choose a size that feels proportional and consistent with your aesthetic goals.
Breast Augmentation Revision Surgery: When and Why It’s Needed
Breast implants are long-lasting but not lifetime devices. Over time, changes such as rupture, capsular contracture, implant shifting, or evolving aesthetic preferences may lead a patient to consider revision surgery.
Revision may involve replacing implants, adjusting their position, removing scar tissue, or combining the procedure with a lift. Because revision can be more complex than initial augmentation, careful evaluation and surgical planning are essential to restore comfort, symmetry, and appearance.
Why Choose Dr. Hayley Brown?
Dr. Hayley Brown is a board-certified plastic surgeon in Las Vegas and Henderson, NV, with more than 25 years of experience performing cosmetic procedures. She operates alongside her husband, Dr. Steve Brown, a board-certified anesthesiologist. Together, they have been working as a team since 1996, combining surgical expertise with the highest standards of patient safety and comfort. All procedures are performed in fully accredited outpatient surgery centers, and Dr. Brown also holds hospital privileges at St. Rose Dominican Siena Hospital.
Patients choose Dr. Brown because of her skill, artistry, and dedication to personalized care. Every detail of care from consultation through recovery is managed with professionalism and compassion.
FAQ
How Often Do Breast Implants Need to Be Replaced?
A common misconception is that breast implants need to be replaced every 10 years. However, they can last up to 15 years, but implants can potentially sustain damage, such as a rupture. If this occurs, you will need to have the implant replaced sooner.
There are signs that your breast implants are ruptured:
- The size of your breasts has changed.
- You feel sudden pain or tenderness in your breasts.
- You feel ripples or lumps in your breasts.
- The shape of your breasts has changed.
If you notice any of these signs, contact Dr. Brown immediately.
What Are the Types of Breast Implants?
There are four different types of breast implants you can get: silicone, saline, gummy bear, and fat graft.
Although each of these options accomplishes the same thing, there are distinct factors that separate them. For example, fat grafting is when fat is taken from another part of your body and placed into your breasts for a fuller appearance. Gummy bear implants are similar to silicone implants, but they also contain the volume of saline implants.
How Long Does Breast Augmentation Surgery Last?
Breast augmentation surgery is usually performed as an outpatient procedure. Patients do have the option to stay overnight as a safety precaution. It is important for patients to arrive at least an hour or two before the procedure begins. As for the procedure itself, breast augmentation surgery usually takes no more than two hours.
How Soon After Childbirth or Breastfeeding Can I Get Breast Augmentation?
It is best to wait at least three to six months after finishing breastfeeding before having breast augmentation. This allows your breasts time to return to their natural size and shape, and gives your body a chance to fully heal. Dr. Brown will evaluate your situation to make sure your results are safe and long-lasting.
Can Weight Gain or Weight Loss Affect My Breast Augmentation Results?
Yes. Significant weight changes can alter the way your breasts look after augmentation. Gaining or losing weight may change your breast size, shape, or skin tightness, which can affect the appearance of your implants. For the most predictable results, it’s recommended to be at or near your goal weight before surgery.
Will I Lose Nipple Sensation After Breast Augmentation?
Some patients experience temporary changes in nipple sensation after surgery, including numbness or increased sensitivity. In most cases, this improves over time as the nerves heal. Permanent changes are possible, but uncommon.
Can Breast Implants Interfere With Mammograms or Breast Cancer Screenings?
Breast implants do not prevent you from getting mammograms, but they can make it more difficult to see all of the breast tissue. Special techniques and extra images are often used to ensure accurate screening. It is important to let the imaging center know you have implants before your appointment.
What Are the Risks of Capsular Contracture, and How Can It Be Prevented?
Capsular contracture happens when scar tissue around the implant tightens, making the breast feel firm or look misshapen. While the exact cause is not always clear, it may be linked to infection, bleeding, or inflammation. Choosing an experienced surgeon, using precise surgical techniques, and following all aftercare instructions can help lower the risk. If capsular contracture does occur, treatment may involve surgery to remove or replace the implant.
Can I Breastfeed After Breast Augmentation?
In most cases, yes. Many women are able to breastfeed successfully after breast augmentation. The ability to nurse depends on factors such as incision location, implant placement, and your natural breast anatomy. Incisions placed in the fold under the breast are less likely to interfere with milk ducts or nerves.
While most patients do not experience long-term issues, breastfeeding success can never be guaranteed, even without implants. If you plan to have children in the future, this should be discussed during your consultation so surgical planning can take that into account.
Can Breast Implants Affect Chest Workouts Long-Term?
After full healing, most women return to their normal exercise routines without limitation. However, very large implants or certain placement techniques may slightly change how chest muscles feel during intense upper body workouts.
Women who lift heavy weights or perform frequent chest-focused exercises should discuss this in advance. Choosing an appropriate implant size and placement can help minimize interference with strength training and maintain comfort during long-term fitness activities.
What Happens If I Decide to Remove My Implants in the Future?
If you choose to remove your implants, a procedure called explant surgery is performed. This may involve simply removing the implants or also removing the surrounding scar tissue, depending on your situation.
After removal, the breasts may appear smaller, looser, or slightly sagging, especially if larger implants were in place for many years. Some patients choose to combine implant removal with a breast lift to reshape and support the natural tissue.
Will I Need Special Bras Permanently After Surgery?
You will need to wear a supportive surgical or underwire bra during the early recovery period to help stabilize the implant position. After healing, most women can wear standard bras, including sports bras and specialty styles.
Permanent “medical” bras are not required, but good support is beneficial for maintaining your results.
How Do I Know If I Need a Lift Instead of Implants Alone?
Breast implants increase volume but do not correct significant sagging. If your nipples sit below the breast crease or point downward, a lift may be necessary to reposition the tissue and create a more youthful contour.
During your consultation, breast position, skin quality, and nipple placement are evaluated to determine whether implants alone will achieve your goals or if combining augmentation with a lift would provide a better result.
Contact Our Breast Augmentation Surgeon
Dr. Hayley Brown understands that every woman has her own vision for how she wants her breasts to look and feel. Whether your goal is a subtle, natural enhancement or a fuller, more dramatic shape, she has the experience and skill to create results that match your preferences. Her artistic eye allows her to help patients feel confident both in and out of clothing.
If you'd like to learn more about your breast augmentation options, please contact us at Desert Hills Plastic Surgery Center using the form on this page or call 702-260-7707 today to schedule a consultation. Dr. Hayley Brown serves patients in Las Vegas, Henderson, and the surrounding areas of Nevada.